 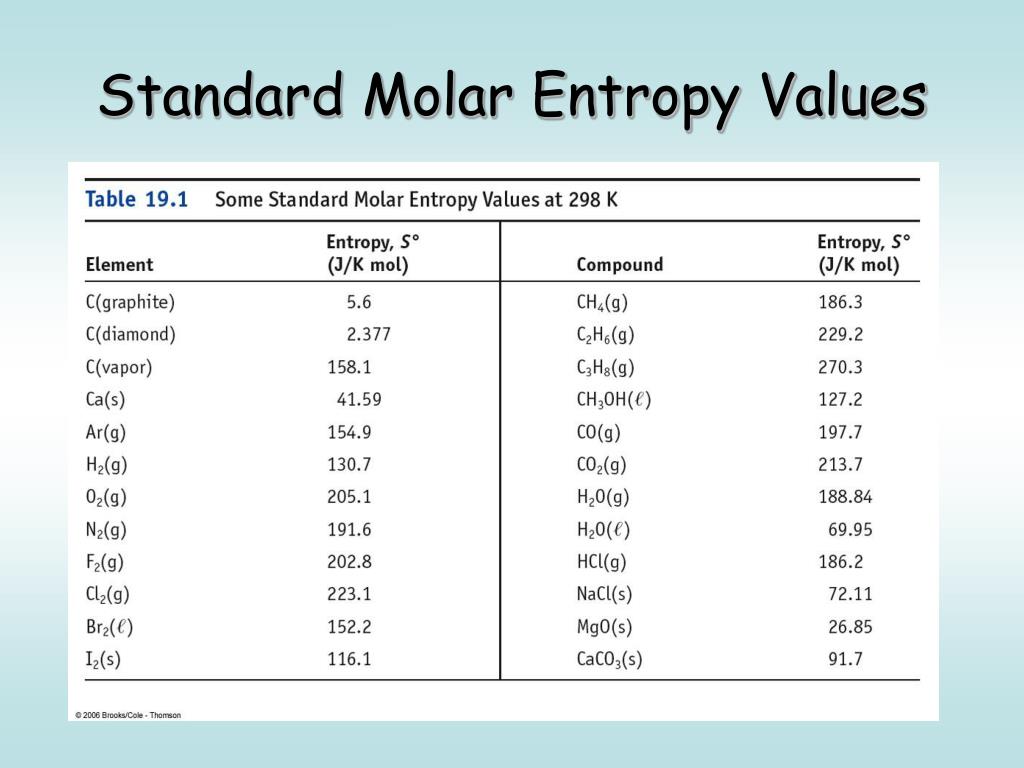
The standard entropy of reaction helps determine whether the reaction will take place spontaneously. ΔS° rxn = S o(products) - S o (reactants) Entropy is a scientific concept, as well as a measurable physical property, that is most commonly associated with a state of disorder, randomness, or uncertainty. Chemical equations make use of the standard molar entropy of reactants and products to find the standard entropy of reaction: The standard molar entropy of a substance is the absolute entropy of 1 mole of the substance in the standard state. Standard molar entropy, S o solid J/(mol K) Heat capacity, c p J/(mol K) Liquid properties Std enthalpy change of formation, f H o liquid-147.6 kJ/mol Standard molar entropy, S o liquid: 229.7 J/(mol K) Heat capacity, c p: 132.42 J/(mol K) 262 ☌ to 3 ☌ Gas properties Std enthalpy change of formation, f H o gas 124.7 kJ/mol. The heat capacity of the gas from the boiling point to room temperature.Ĭhanges in entropy are associated with phase transitions and chemical reactions. 
The latent heat of vaporization of the liquid. Title, Excess partial molar entropy of alkane-mono-ols in aqueous solutions at 25 degrees C. Iron oxide (or rust) is a spontaneous reaction that occurs at a very slow rate in usual atmospheric conditions.The heat capacity of the liquid from the melting point to the boiling point.The latent heat of fusion of the solid.The heat capacity of one mole of the solid from 0 K to the melting point (including heat absorbed in any changes between different crystal structures).19, we obtain the following expression for the molar. (20) S S v + S t + S r Substituting the vibrational partition function given in Eq. The standard molar entropy of a gas at STP includes contributions from: The entropy is given by : (19) S k B l n Q + k B T ( l n Q V) T The molar entropy is a combination of three contributions, including vibrational, translational and rotational entropies. The total molar entropy is the sum of many small changes in molar entropy, where each small change can be considered a reversible process. Here, dq k/T represents a very small exchange of heat energy at temperature T. Liquid phase molar entropy at standard conditions, 1 bar standard pressure (S° liquid,1 bar). The total electronic heat capacity for figure 1 and figure 2 is (26) The contribution of the partial molar electronic entropy to the total electronic entropy in Figure 1 is (27) The partial molar electronic heat capacity contributes only 28.46 of the total electronic heat capacity. Under identical conditions, it is greater for a heavier gas.If a mole of substance were at 0 K, then warmed by its surroundings to 298 K, its total molar entropy would be the addition of all N individual contributions: Molar entropy is not the same for all gases. White Rose Research Online URL for this paper. The standard molar entropy at pressure = P 0 This is a repository copy of Estimation of standard molar entropy of cement hydrates and clinker minerals. These are often (but not necessarily) chosen to be the standard temperature and pressure. 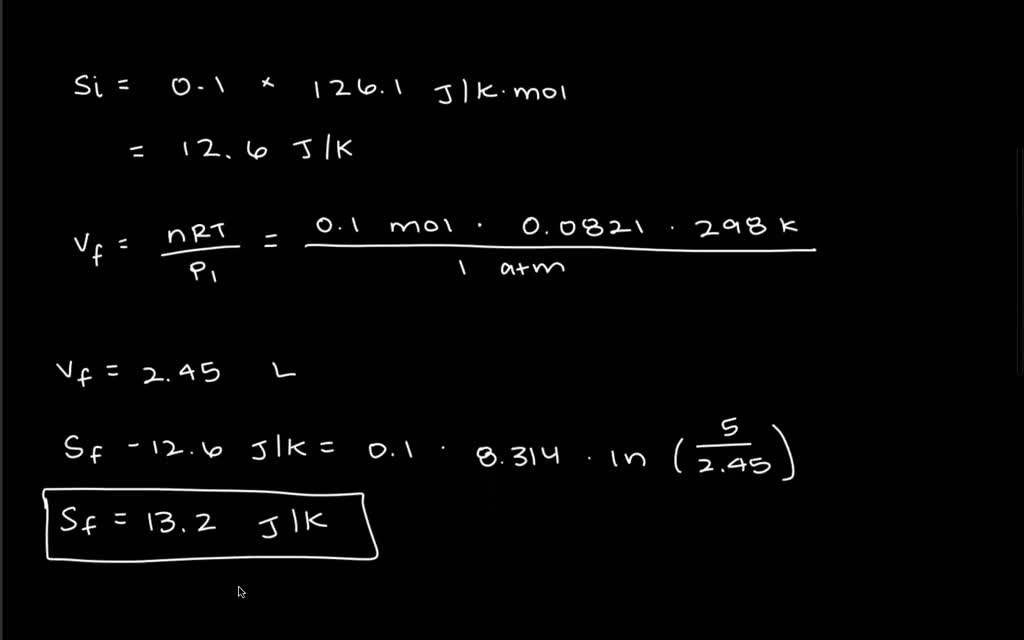 
Sizes and shapes add to the complexity of a molecule. The more complex the molecule is, the greater the standard molar entropy. Unlike enthalpy or internal energy, it is possible to obtain absolute entropy values by measuring the entropy change that. In chemistry, the standard molar entropy is the entropy content of one mole of pure substance at a standard state of pressure and any temperature of interest. Standard molar entropy is the entropy (likelihood or probability that the system will be in a particular state) of one mole of a substance under a standard state of conditions. One way of calculating S for a reaction is to use tabulated values of the standard molar entropy (S°), which is the entropy of 1 mol of a substance at a standard temperature of 298 K the units of S° are J/(mol·K). Standard entropy content of one mole of a substance under a standard state 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
